On October 15, the U.S. FDA granted orphan drug designation for APX005M—a CD40 immunomodulator—for the treatment of pancreatic cancer as well as esophageal and gastroesophageal junction cancer. This is the same immunomodulator used in the CRI Clinical Accelerator’s PRINCE trial, which is exploring new treatments for metastatic pancreatic cancer. This designation will help drive further drug development and hopefully, more promising treatments for this devastating disease.
This November for Pancreatic Cancer Awareness Month, we look at new research, new treatments, and how we’re working toward a future immune to pancreatic cancer.
Pancreatic Cancer Treatment Landscape Update
At the 2019 CRI Immunotherapy Patient Summit in Baltimore, Elizabeth M. Jaffee, MD, deputy director of the Sidney Kimmel Comprehensive Cancer Center, shared research and treatment news for pancreatic cancer.
Ask Your Immunotherapy Questions
Join Dr. Jaffee for a special breakout session on pancreatic cancer at the CRI Virtual Immunotherapy Patient Summit on December 5. This free event connects you with cancer patients and immunotherapy experts, all from the comfort and safety of your own home.
 Pancreatic Cancer Scientist Spotlight
Pancreatic Cancer Scientist Spotlight
CRI CLIP Investigator Vincenzo Bronte, MD, has shed light on a new class of proteins involved in T cell homing and retention within a tumor. Now, he aims to characterize how these proteins interact with both tumor and immune cells in order to uncover new immunotherapy solutions for patients with pancreatic cancer.
Learn About Dr. Bronte’s Research
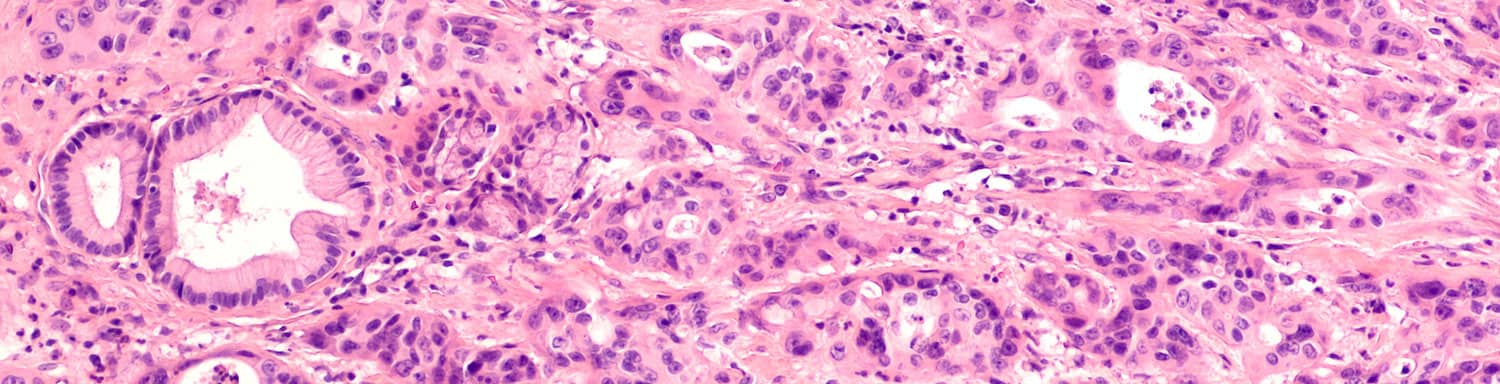
 Immunotherapy for Pancreatic Cancer
Immunotherapy for Pancreatic Cancer
Information Updated
On June 16, 2020, the U.S. FDA approved pembrolizumab (Keytruda), a PD-1 checkpoint inhibitor, to treat adult and pediatric patients whose advanced solid cancers—including pancreatic cancer—have a high tumor mutational burden (TMB-H) and have proven resistant to available treatments. This is the second biomarker-based indication regardless of cancer type to receive FDA approval. As the research and treatment landscape changes, we keep our immunotherapy for pancreatic cancer information up to date.
 Find a Pancreatic Cancer Clinical Trial
Find a Pancreatic Cancer Clinical Trial
A variety of new and promising cancer immunotherapy treatments are only available to patients in clinical trials. Our Immunotherapy Clinical Trial Finder will match you to trials for which you may be eligible, and you can help speed the development of potentially lifesaving drugs for yourself and others.
Support Pancreatic Cancer Research
Robert Vonderheide, MD, DPhil, a member of CRI’s Scientific Advisory Council, is leading a CRI clinical trial that is targeting the CD40 pathway in combination with chemotherapy and checkpoint immunotherapy in patients with metastatic pancreatic cancer. Early results have demonstrated the promise of this approach. This Pancreatic Cancer Awareness Month, support lifesaving cancer immunotherapy research.