2017 has been quite a year for immunotherapy. In February, Immunotherapy 2.0 was declared the clinical advance of the year by the American Society of Clinical Oncology for the second straight year. Since then, there have been over a dozen cancer immunotherapy approvals, including the first bispecific T cell engaging (BiTE) antibody, the first two CAR T cell immunotherapies, and the first treatment of any type to be approved for solid tumors that have a specific genomic biomarker regardless of where they’re located.
Accordingly, immunotherapy is now featured prominently at major conferences and has even spawned new conferences focused exclusively on immunotherapy. Two conferences—one in America and one in Europe—took place this past weekend.
From December 7-10 in Geneva, Switzerland, the European Society for Medical Oncology held their inaugural Immuno Oncology Congress 2017 (ESMO IO), while the American Society of Hematology’s (ASH) annual meeting in Atlanta, Georgia, took place December 10-12, and important contributions by Cancer Research Institute (CRI) scientists were highlighted at both.
ESMO IO
At ESMO IO, talks covered diverse immunotherapy insights and approaches. Most notably, George Coukos, MD, PhD, gave the keynote lecture on how the field has come to appreciate the impact of the tumor microenvironment as well as how that can help inform therapeutic approaches for patients, while Aiman Shalabi, Pharm.D., MBA, BCOP, discussed the role of venture philanthropy in fueling innovation in the cancer immunotherapy space. Shalabi’s talk focused especially on a recent publication by CRI’s Clinical Accelerator team, which represented first-of-its-kind analysis of the global immunotherapy clinical landscape from pre-clinical development to regulatory approval.
Ignacio Melero, PhD, a currently funded CRI CLIP Investigator, gave a talk about “what’s new” in liver cancer immunotherapy. Two posters also featured his work: one highlighted the combination of a dendritic cell vaccine and radiotherapy in advanced cancer patients, while the other revealed that expression of the LAG-3 immune checkpoint predicted resistance to anti-PD-1 immunotherapy in patients with advanced non-small cell lung cancer (NSCLC). Sergio A. Quezada, PhD— previously funded by CRI from 2005-2008 and 2011-2015—spoke about clonal neoantigens, an important area of work in cancer immunotherapy that has the potential to provide personalized, patient-specific approaches. Additionally, a poster covered Dr. Quezada’s efforts to determine how the T cell receptor (TCR) repertoires of non-small cell lung cancer (NSCLC) patients affect risk of recurrence.
A poster showcased the work of Dirk Jaeger, MD—previously funded by CRI from 1998-2000 and 2006-2010—involving a first-in-human clinical trial in which melanoma patients have been treated with novel mRNA-lipoplex nanoparticles that stimulate immune responses targeting shared tumor antigens. Lastly, Ira Mellman, PhD, explored combination cancer immunotherapy strategies and emphasized the importance of basing combinations on sound scientific mechanisms, while Kunle Odunsi, MD, PhD– previously funded by CRI from 2011-2014 and 2015-2016—discussed immunotherapy opportunities in ovarian cancer.
ASH 2017
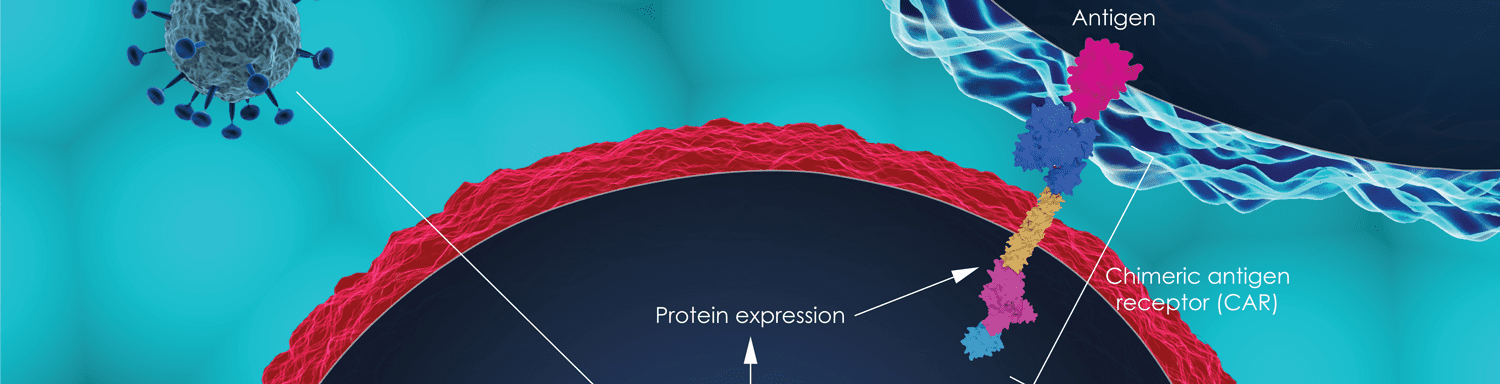
While ESMO IO’s focus was immunotherapy in general, ASH highlighted immunotherapy breakthroughs in blood cancers such as leukemia, lymphoma, and multiple myeloma. Due to the two recent CAR T cell immunotherapy approvals in leukemia and lymphoma, many sessions and talks at ASH discussed the updated results from the relevant CAR T cell clinical trials and a few others. By and large, the updates from these anti-CD19 CAR T cell trials in leukemia and lymphoma showed that the majority of the patients who respond to these treatments remained in remission throughout their follow up periods. One strategy even demonstrated an improved safety profile after treatment, which led them to suggest that this approach could potentially be administered in an outpatient setting in the future.
Further breakthroughs in the realm of CD19-targeting CAR T cell immunotherapies included work involving Carl H. June, MD, and Michael Kalos, PhD, who highlighted the case of a patient with chronic lymphocytic leukemia (CLL) who responded to CAR T cell immunotherapy in a unique and insightful way. Through a random mutation, a gene known as TET2 became de-activated in one of the billions of CAR T cells that the patient received, making that clone—and all of its “descendants”—particularly effective at seeking out and eliminating the patient’s leukemia cells. Additional work involving June focused on the benefits experienced by a lymphoma patient who received anti-PD-1 checkpoint immunotherapy after initial disease progression following CAR T cell treatment. Other CD19-based CAR T cell talks and posters covered everything from biomarkers of toxicity (via Stanley R. Riddell, MD) and efficacy (via E. John Wherry, PhD), in addition to model-based analysis (via June) and “off-the-shelf” CAR T cell products (via Michel Sadelain, MD, PhD). Another interesting approach that was tested in mice revealed the effectiveness of CAR T cells that secreted their own PD-1 blocking molecules. Checkpoint immunotherapies targeting the PD-1 pathway enable T cells to stay activated as they target and eliminate cancer cells, but unfortunately can also cause immune reactions against healthy cells. This strategy localized the PD-1-blocking effects to the tumor regions and thus limited off-target effects.
Beyond CD19-targeting approaches, June was also involved in approaches using CAR T cells to target BCMA (B cell maturation antigen) in multiple myeloma patients as well as CD22 in acute lymphoblastic leukemia (ALL). Additionally, Markus Muschen, MD, PhD, a currently funded CRI CLIP Investigator, analyzed different pre-ALL subtypes and was able to identify novel subtype-specific regulatory components that influence the disease. Other treatments highlighted in multiple myeloma were a CAR T cell immunotherapy that simultaneously targeted both BCMA and CD38 in multiple myeloma, a BCMA-targeting antibody-drug conjugate (via Axel Hoos, MD, PhD), and bispecific T cell-engaging antibodies that targeted CD33 and CD38 in mice.
ASH 2017 also showcased a number of preclinical insights that could potentially be exploited to improve immunotherapy’s effectiveness in the clinic. Among these were an approach involving dendritic cell expansion that was shown to provide a chemotherapy alternative (that prevented graft-versus-host disease) after bone marrow transplants (via Mark John Smyth, PhD, FAHMS) as well as a strategy involving venetoclax-mediated inhibition of natural killer cells that allowed for less intense chemotherapy after stem cell transplantation in mice with acute myeloid leukemia (via Nicholas D. Huntington, PhD). Importantly, venetoclax treatment improved stem cell engraftment and prevented graft-versus-host disease without sacrificing the anti-leukemia benefits. Similarly, inhibition of lutenizing hormone (LH) was shown to improve the effectiveness of stem cell transplantation after radiation (via Marcel van den Brink, MD, PhD). Finally, various studies involving Markus Muschen, MD, PhD, revealed the significance of autoimmunity checkpoints—including LRG5, CD25, BCL6, PON2—that play a role in B cell malignancies and the potential promise of targeting them therapeutically in patients.
These two conferences showcased a wide variety of advances that have been made in immunotherapy recently, and there’s great reason to believe that immunotherapy’s benefits will keep on improving for patients. To learn more about what’s next in immunotherapy, be sure to watch our “Immunotherapy: A Glance at the Year Ahead” webinar with Jedd D. Wolchok, MD, PhD, on January 16, 2018.