If you or a loved one has been diagnosed with cancer, you’ve likely heard the term “clinical trial”. But what does it really mean — and could it be an option for you?
If you or a loved one is interested in exploring clinical trials, getting started often begins with a conversation. Speaking with a doctor can help determine whether a trial might be a good fit. It’s also important to consider personal goals, preferences, and practical factors like time, travel, and support systems. Asking questions — and taking the time to understand all options — can make the process feel more manageable.
Importantly, no one has to navigate this process alone. Clinical trial navigators and support services like CRI’s Clinical Trial Finder are available to help patients and families understand their options and take the next steps with confidence:
Understanding Cancer Clinical Trials
Cancer clinical trials are research studies designed to test new ways to prevent, detect, or treat cancer. They are a critical part of how progress happens in cancer care. In fact, every treatment that is now considered “standard of care” was once studied in a clinical trial.
Clinical trials vary widely in their purpose. While many focus on testing new treatments, others aim to prevent cancer, improve screening and diagnosis, or enhance quality of life by reducing symptoms and side effects. Some may focus on entirely new therapies, including innovative approaches like immunotherapy. Others look to expand existing treatments to more people.
The many different types of clinical trials reflect the many ways research is working to improve both outcomes and patient experience.

“Every miraculous breakthrough has come from clinical trials.”
– CRI STAR Joshua Brody, MD, Icahn School of Medicine at Mount Sinai
Why Clinical Trials Matter
Clinical trials play a vital role in advancing research, but they can also offer meaningful options for patients today. Participation may provide access to treatments that are not yet widely available, or new approaches when standard therapies haven’t worked as hoped. Patients in clinical trials are also closely monitored by specialized care teams, often with additional support throughout their treatment journey.
Beyond individual benefit, every person who participates contributes to research that could improve outcomes for future patients.
“I began to look at clinical trials, and it wasn’t only that I was fighting for my life, but I really wanted to pay it forward, help cancer research, and help other cancer patients, even if it wasn’t going to work for me.”
– Kristin Kleinhofer, Stage 4 Leukemia Survivor

Despite their importance, clinical trials are often misunderstood. One of the most common misconceptions is that they are only a “last resort.” In reality, clinical trials are available at many points along the treatment journey. Depending on the specific diagnosis and treatment plan, a trial may be an option even early in care.
Regardless of when you might be eligible for a clinical trial, it is worth asking the questions. Watch below to learn how a clinical trial changed the lives of these once-terminal cancer patients.
The Clinical Trial Process
To ensure safety and effectiveness, clinical trials are conducted in phases, with key considerations for each phase.
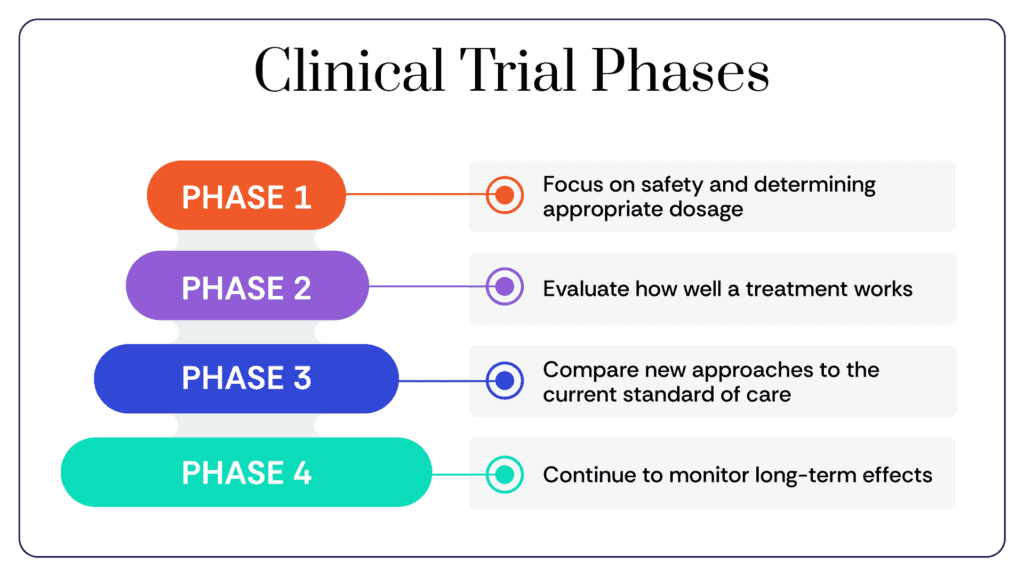
Each phase builds on the knowledge gained from the previous one. Not all trials make it through all 4 phases.
Clinical Trial Considerations
Like any treatment decision, joining a clinical trial involves weighing potential benefits and risks.
Some patients may gain access to promising new therapies and receive close medical oversight while also contributing to research that helps others. At the same time, side effects can occur – just as they can with standard treatments – along with additional visits or testing, and there is no guarantee that the new treatment will be more effective than existing options. These are important considerations to discuss with the clinical trial team.
Similarly, not every clinical trial is the right fit for every patient. Each study has specific eligibility criteria based on factors such as cancer type and stage, prior treatments, overall health, and sometimes biomarkers or genetic features. These criteria are designed to protect patients and ensure that researchers can accurately understand how a treatment works.
Cost is another common question. In many cases, the trial sponsor covers the cost of the study treatment and related procedures. However, routine care — such as standard doctor visits — may still be billed to insurance. It’s always helpful to clarify with your plan what is covered before deciding to participate.
A Path Toward Possibility
Clinical trials remain a cornerstone of cancer research — and for many patients, they represent not just progress, but possibility. Whether or not participation is the right choice, understanding how clinical trials work can empower patients to make informed decisions about their care.
Sometimes, exploring a clinical trial starts with a simple question. And that question can open the door to new possibilities.

“Clinical trials are the lifeline for long-term survival rates with cancer. There is no other way to move this field forward, for patients and for everybody else, that would produce any kind of meaningful change. If it wasn’t for clinical trials, I wouldn’t be here.”
– Sharon Belvin, Stage 4 Melanoma Survivor
10 Questions to Ask Your Doctor About Clinical Trials
If you’re considering a clinical trial, having an open conversation with your doctor is an important first step. Bringing a list of questions can help you feel more prepared, confident, and informed.
You can also print this page and use it as a guide during your appointment.
Are there any clinical trials that might be right for me?
Ask whether trials are available based on your specific diagnosis, stage, markers, and treatment history.
What is the goal of this clinical trial?
Is the trial testing a new treatment, a combination of therapies, or something else, like quality of life or managing symptoms?
How does this trial treatment compare to my current or standard treatment options?
Understanding the differences can help you weigh whether a trial is the right path.
What are the possible benefits for me?
What outcomes are researchers hoping for?
What are the potential risks or side effects?
Make sure you understand both known side effects and any unknowns.
What phase is the clinical trial in?
Knowing whether it’s an early- or late- phase trial can give you insight into how much is already known about the treatment and experience.
How long is the trial, and what would participation involve on a day-to-day basis?
Ask about time commitment, number of visits, tests, and any travel requirements.
What happens if the treatment doesn’t work for me?
Will I have other options, and can I leave the trial?
What costs are covered, and what will I or my insurance need to pay?
Understanding the financial aspect upfront can help avoid surprises.
How will this trial affect my quality of life?
This includes side effects, time demands, and how treatment may impact your daily routine.
