-

How Man’s Best Friend Could Help Us Cure Cancer
Cancer very is similar in dogs and humans, and scientists are now exploring how cures for one…
-
Immunotherapy is Here to Stay: Looking Back at this Year’s Breakthroughs
Important immunotherapy breakthroughs benefited patients immensely in 2017 and paved the way for even more advances in…
-

Recent FDA Announcements Herald Advances in Personalized Approaches Against Cancer
Two tumor-profiling tests recently authorized by the FDA enable doctors to tailor their treatments more effectively to…
-
Immunotherapy Shines at ASH and ESMO IO Conferences
Two conferences, one in America and one in Europe, highlight immunotherapy's continued progress and the important contributions…
-
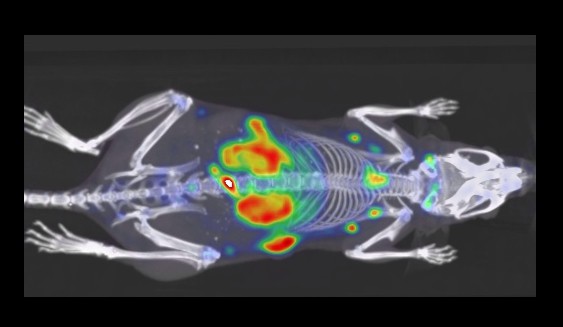
CRI Researcher Highlights New Technique to Aid Immunotherapy
Dr. Mohammad Rashidian’s nanobodies could help improve doctors’ decisions regarding immunotherapy.
-
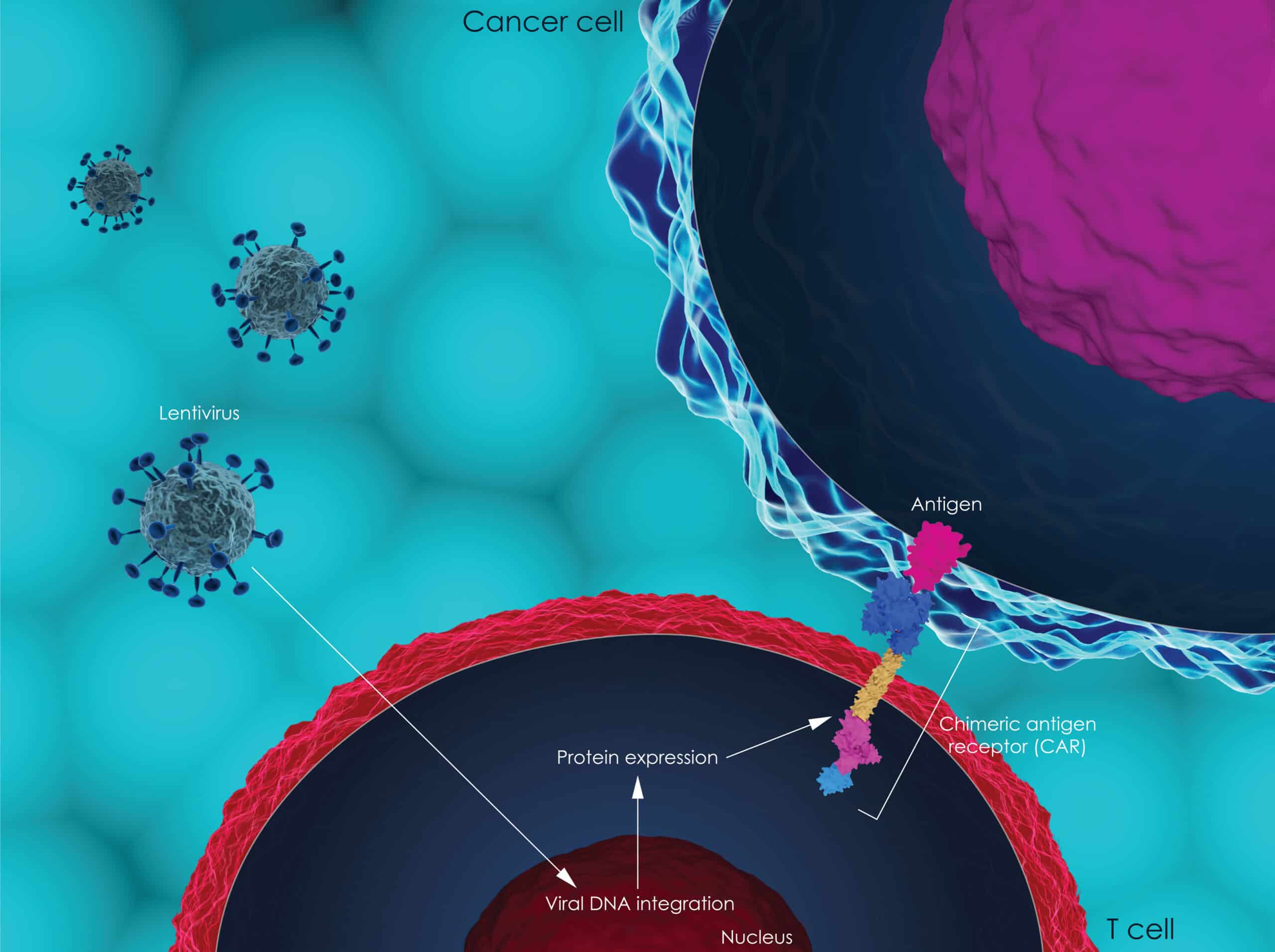
CAR T Cell Immunotherapy Approved for Adult Non-Hodgkin Lymphoma Patients
FDA approves new immunotherapy–Yescarta (axicabtagene ciloleucel)–for adult patients with relapsed or refractory non-Hodgkin large B cell lymphoma.
-

Checkpoint Immunotherapy Approved for Patients with Stomach, Gastroesophageal, and Liver Cancer
Previously treated patients with these cancer types can now receive anti-PD-1 immunotherapy; nivolumab for HCC and pembrolizumab…
-
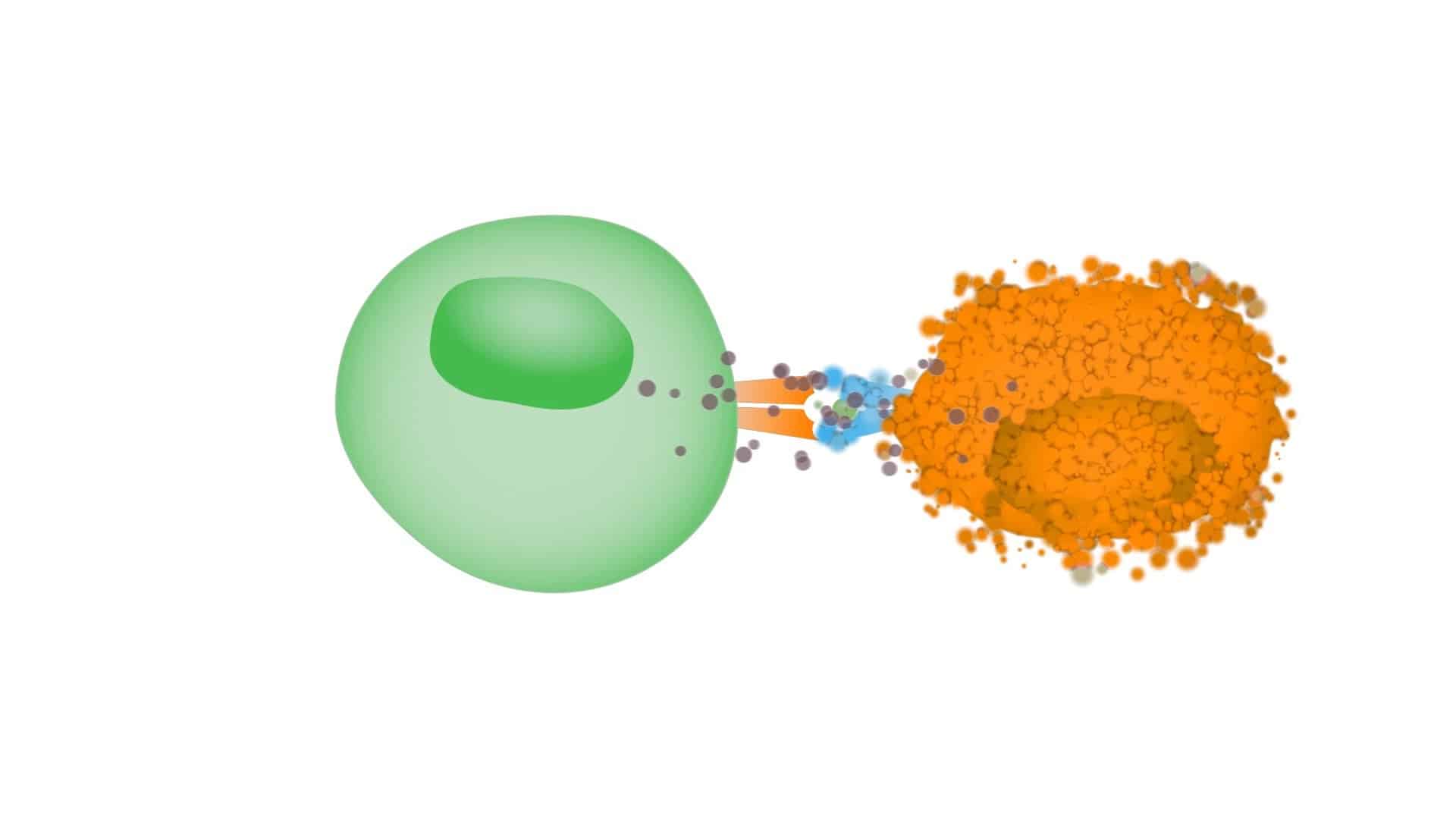
CICON17 Day 4 Recap: The Tumor Microenvironment and Oncolytic Viruses
The final day of CICON17 explored two newer immunotherapy approaches that reflect broader understanding of tumor-immune interactions…
-

CICON17 Day 3 Recap: Combinations, Checkpoint Immunotherapy Resistance, Immunomodulators, and Microbiota
Day 3 of the CICON17 focused on combination therapies, immunomodulators, overcoming immune suppression, and the role bacteria in…